UK regulators have issued a warning that people who have a history of "significant" allergic reactions should not currently receive the Pfizer/BioNTech Covid-19 vaccine after two people who had the jab yesterday had allergic reactions.
Two NHS staff members who received the vaccine on the first day of the mass vaccination programme suffered an allergic reaction, the NHS in England has confirmed.
The Medicines and Healthcare products Regulatory Agency (MHRA) has given precautionary advice to NHS trusts that anyone who has a history of "significant" allergic reactions to medicines, food or vaccines should not receive the vaccine.
The NHS in England said all trusts involved with the vaccination programme have been informed.
This means that anyone scheduled to receive the vaccine today will be asked about their history of allergic reactions.
Professor Stephen Powis, national medical director for the NHS in England, said: "As is common with new vaccines, the MHRA have advised on a precautionary basis that people with a significant history of allergic reactions do not receive this vaccination after two people with a history of significant allergic reactions responded adversely yesterday.
"Both are recovering well."
The MHRA advice states: "Any person with a history of a significant allergic reaction to a vaccine, medicine or food (such as previous history of anaphylactoid reaction or those who have been advised to carry an adrenaline autoinjector) should not receive the Pfizer/BioNtech vaccine.
"Resuscitation facilities should be available at all times for all vaccinations. Vaccination should only be carried out in facilities where resuscitation measures are available."
It is understood that both the staff members had a significant history of allergic reactions, to the extent where they need to carry an adrenaline auto injector with them.
They developed symptoms of "anaphylactoid reaction" shortly after receiving the vaccine and both have recovered after the appropriate treatment.
Pfizer said the vaccine was "well tolerated" during the trials with "no serious safety concerns".
A spokeswoman said: "We have been advised by MHRA of two yellow card reports that may be associated with allergic reaction due to administration of the Covid-19 BNT162b2 vaccine.
"As a precautionary measure, the MHRA has issued temporary guidance to the NHS while it conducts an investigation in order to fully understand each case and its causes. Pfizer and BioNTech are supporting the MHRA in the investigation.
"In the pivotal phase three clinical trial, this vaccine was generally well tolerated with no serious safety concerns reported by the independent Data Monitoring Committee.
"The trial has enrolled over 44,000 participants to date, over 42,000 of whom have received a second vaccination."
England's chief medical officer Prof Chris Whitty has said that the NHS and the MHRA are in a "very good position" to pick up "extremely rare" issues with any vaccine once it is in use.
Thousands of Britons became the first in the Western world to received an approved Covid vaccine yesterday as the NHS began the biggest vaccination drive since it was created in 1948.
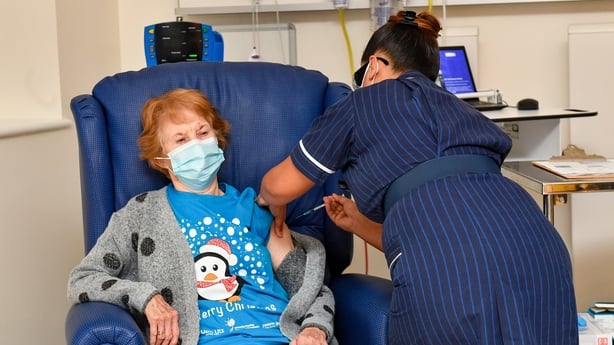
The vaccine is administered in two doses, 21 days apart. The over-80s and health and social care staff are first in line to get the jab in the national roll-out.
Britain has received some 800,000 doses of the vaccine in the first batch of an order of 40 million. Up to four million doses are expected by the end of December.
Pfizer chief executive Albert Bourla yesterday said he understood global concerns about the speed with which pharmaceutical companies have produced vaccines against Covid-19.
But he insisted no corners have been cut.
The vaccine had been tested "in the exact same way as we are testing any vaccine that is circulating out there", he told a virtual media briefing in Geneva.
Additional reporting AFP