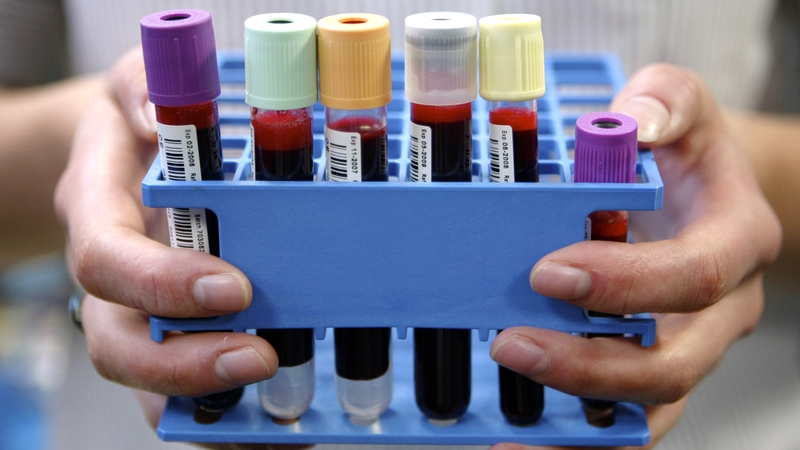
Analysis: a blood cancer biobank is an invaluable tool for researchers looking for better treatments to combat these diseases
Blood cancers such as acute myeloid leukaemia (AML), multiple myeloma (MM) and chronic lymphoid leukaemia (CLL) are very rare and complex diseases. They vary greatly between patients and are very different at the genetic, cellular and cellular microenvironment levels. While some advances have been made over the last decades in treatment, many of these cancers still remain difficult to treat.
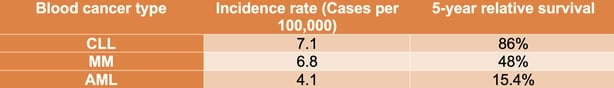
According to the National Cancer Registry Ireland (NCRI), almost 700 people on average are diagnosed every year with blood cancers. The prognosis for survival varies a lot (see table below), but the haemotology research community continues to work on improving the diagnosis, treatment and quality of life for patients with these sometimes-aggressive diseases.
A lot of research is required in order to come up with better treatments for blood cancers. This involves screening the proposed treatment on a large set of patient samples to find out whether it is widely effective. Typically, the number of patient samples one can obtain is a bottleneck for these studies, as the incidence of these cancers is rare.
Blood Cancer Network Ireland has alleviated some of that problem for haematology researchers in Ireland by setting up a national blood cancer biobank. A biobank is a collection of biospecimens, such as biopsies from tissues. In the case of the blood cancer biobank, it is a collection of bone marrow and blood samples from blood cancer patients.
The samples that come into the biobank first need to be processed to isolate the fractions that the biobank stores, which is done by highly trained staff. Many quality control steps are undertaken to ensure that the samples stored are of the highest quality possible. All procedures carried out are well documented and are fully GDPR compliant.
We need your consent to load this rte-player contentWe use rte-player to manage extra content that can set cookies on your device and collect data about your activity. Please review their details and accept them to load the content.Manage Preferences
From RTÉ Radio 1's Ray D'Arcy Show during Blood Cancer Awareness Month 2019, Ciaran Byrne talks about being diagnosed with leukemia
The biobanking process starts when a clinician suspects a patient of having a blood cancer. The patient is asked to donate a sample for the biobank. If the patient consents, a sample of bone marrow and/or peripheral blood is taken for the biobank alongside their clinical sample and a biobank code is generated. Samples can also be collected from the same patient after the completion of treatment and at relapse (if it occurs). This allows for the monitoring of disease progression in the same patient on a molecular and cellular level.
The sample assigned for the biobank is then taken to the biobank laboratory, where the range of fractions that are stored within the biobank get isolated from the whole bone marrow and/or peripheral blood. These include the blood plasma, mononuclear cells (leukaemic cells) and bone marrow stromal cells. These are then stored in ultra-low temperatures until needed by researchers. These samples are stored in special tubes which are labelled with individual QR codes.
Plasma isolated from blood can be used to identify biomarkers, which may be used to predict disease severity or treatment responses. The isolated mononuclear cells (leukaemic cells) can be used to evaluate the efficacy of new therapies. Finally, stromal (or supporting) cells can be used to understand the cancer cell environment and how those cells might help cancer cells to evade the anti-cancer effects of the immune system.
From Blood Cancer Network Ireland, Dr Eva Szegezdi on how the biobank works
The stored samples also include such biospecimen data as concentration of the cells frozen down, the number of tubes frozen down and the tubes QR codes. Some basic clinical data is also recorded such as the precise diagnosis, the treatment details and the response to the treatment.
Any researcher with an ethically approved study can apply for the use of biobank samples. The samples can support research either through prospective sample collection (where a researcher requests specific samples to be collected and stored until required) or through the use of already collected samples.
There are samples from over 500 patients at different stages of the disease currently stored at the Blood Cancer Biobank. These 15,000 tubes were collected by seven different hospitals across Ireland who are part of biobank. If a researcher is looking for a specific subtype of a blood cancer or patients that received a certain kind of treatment, the biobank should be a choice of samples for that study.
The blood cancer biobank is an invaluable tool for the haematology community in Ireland
One of the studies which the biobank currently supports examines the response of chronic lymphocytic leukaemia patients to Covid-19 vaccines. Due to impaired B cell production, CLL patients are immunocompromised and are known to have incomplete response to immunisation. The aim of the study is to understand better the actual efficacy of Covid vaccination in these patients and whether booster vaccines can improve it.
The blood cancer biobank is an invaluable tool for the haematology community in Ireland. It provides a solution to conducting more thorough studies to improve treatment options for future blood cancer patients than otherwise would be possible in a short timeframe. This, in turn, will increase the survival rate for blood cancer patients.
The views expressed here are those of the author and do not represent or reflect the views of RTÉ